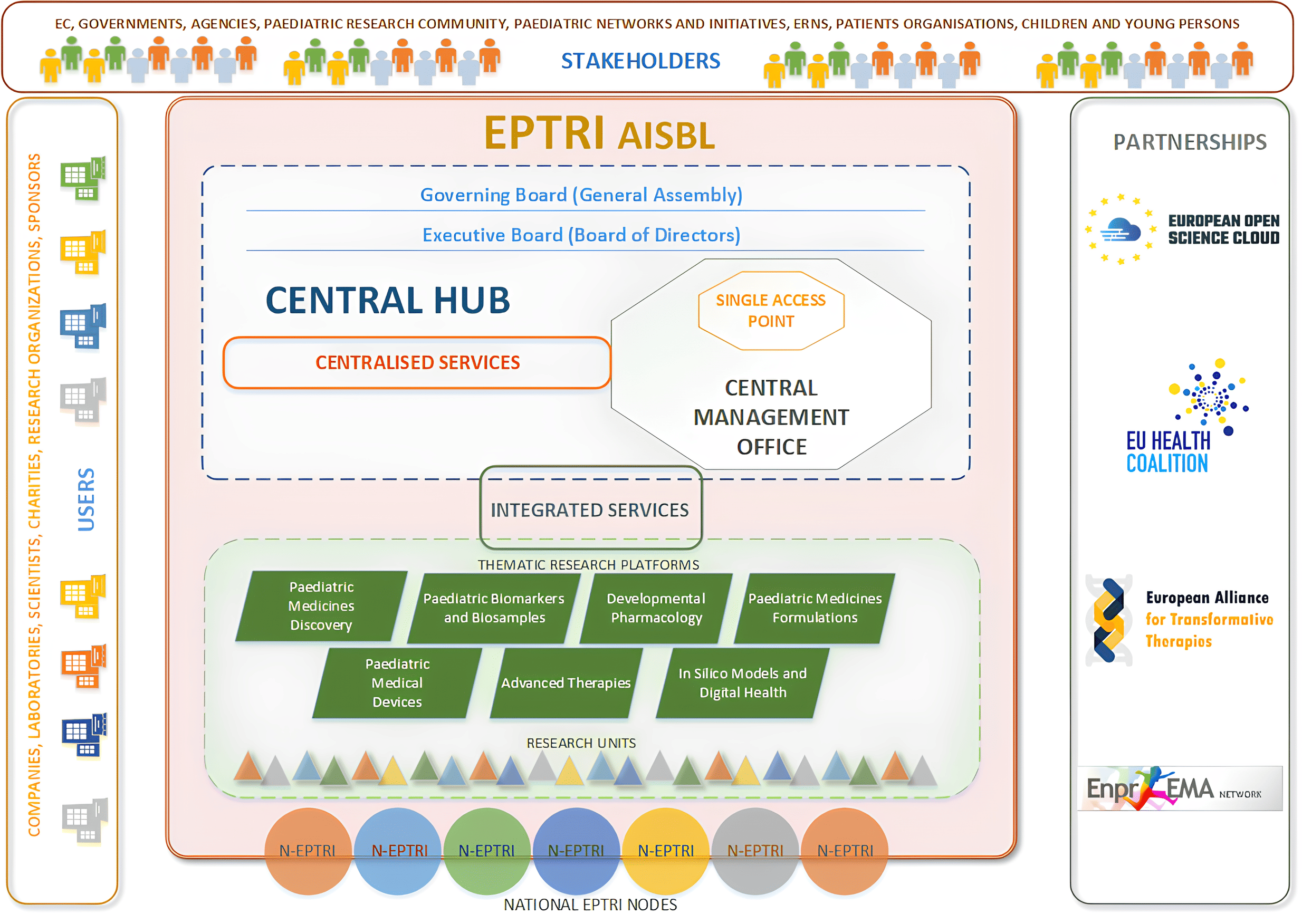
The European Paediatric Translational Research Infrastructure (EPTRI), started as an EU-supported initiative (EU-EPTRI-ID n. 777554), is a distributed Research Infrastructure (RI) composed of several research units grouped both within Thematic Research Platforms – TRPs (according to the field of expertise) and National Nodes (according to their location).
EPTRI is a non-profit research organisation incorporated in the form of an Association Internationale Sans But Lucrative (AISBL) governed by Belgian law, based in Leuven.
Our Statutes are available HERE.
EPTRI is an open science space allowing researchers to work together without geographical, institutional, or financial barriers and a system of many interconnected research areas, that will contribute to bringing new paediatric medicines on the market for the benefit of children.
EPTRI promotes processes allowing stakeholders’ engagement and knowledge translation by integrating the perspectives of young patients and families, policymakers and payers, regulators and researchers, agencies and charities, academia and industry.